Understanding Trichloroetano: From Industrial Solvent to Regulated Substance

Trichloroetano, specifically 1,1,1-trichloroethane (also known as methyl chloroform), has a rich history intertwined with industrial progress and environmental awareness. Its journey from widely used solvent to a regulated substance under the Montreal Protocol highlights the complexities of balancing technological advancement with environmental responsibility. This article delves into the properties, applications, and eventual phase-out of this once-ubiquitous chemical.
The Chemistry and Production of 1,1,1-Trichloroetano
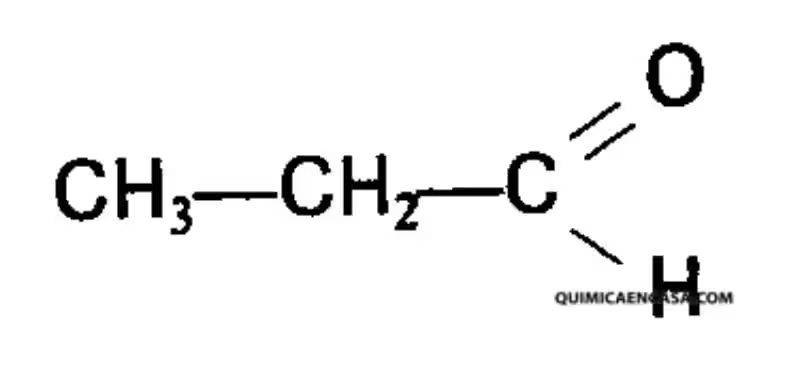
1,1,1-Trichloroetano is a colorless liquid with a characteristically sweet odor. Its chemical formula, CH₃CCl₃, reveals its structure: a methane molecule (CH₄) with three of its hydrogen atoms replaced by chlorine atoms. This seemingly simple substitution has profound effects on its properties, making it a powerful solvent.
The industrial production of 1,1,1-trichloroetano typically involves a two-step process. First, vinyl chloride reacts with hydrogen chloride to yield 1,1-dichloroethane. This intermediate then undergoes a chlorination reaction under ultraviolet (UV) light, resulting in the formation of the final product—1,1,1-trichloroetano. A less common method involves the reaction of 1,1-dichloroethene with hydrogen chloride. The final product frequently contains stabilizers (up to 8%) to inhibit degradation and prevent metal corrosion. These additives help extend the shelf life and usability of the solvent.
Widespread Applications and Historical Use
Before its regulation, 1,1,1-trichloroetano enjoyed widespread use due to its excellent solvency properties and relatively low toxicity compared to other chlorinated hydrocarbons. Its versatility led to numerous applications across diverse industries.
- Metal Cleaning: It was extensively used for cleaning metal parts and electronic circuit boards, effectively removing grease and other contaminants.
- Electronics Manufacturing: In the electronics industry, 1,1,1-trichloroetano served as a photoresist solvent, playing a crucial role in the fabrication of integrated circuits.
- Aerosols and Cutting Fluids: It found application as an aerosol propellant and an additive in cutting fluids, improving their performance and efficiency.
- Other Applications: The list extends to inks, paints, adhesives, coatings, dry cleaning of leather and suede, photographic film cleaning, and even as a thinner in correction fluids. Early exploration also included its potential as an anesthetic and veterinary anthelmintic, though neither application gained widespread use.
Health and Environmental Concerns
Despite its relatively low toxicity compared to some other chlorinated solvents, 1,1,1-trichloroetano is not without risks. It acts as a central nervous system depressant, potentially causing dizziness, confusion, unconsciousness, and even death at high concentrations. Prolonged skin contact can lead to irritation. Furthermore, the International Agency for Research on Cancer (IARC) classifies 1,1,1-trichloroetano as a Group 2A probable carcinogen, meaning there is limited evidence of its carcinogenicity in humans but sufficient evidence in experimental animals.
The major environmental concern associated with 1,1,1-trichloroetano is its ozone-depleting potential. This property led to its phase-out under the Montreal Protocol, an international treaty aimed at protecting the ozone layer. While possessing a relatively short atmospheric lifetime (around 5 years), its ozone depletion potential and its status as a potent greenhouse gas (GWP of 169 relative to CO₂) necessitated its eventual ban.
The Montreal Protocol and the Phase-Out of Trichloroetano
The Montreal Protocol, initiated in 1987, set in motion a phased reduction and eventual elimination of numerous ozone-depleting substances, including 1,1,1-trichloroetano. The phase-out, beginning in 1996, significantly reduced its atmospheric concentration. This led to a search for and implementation of safer, more environmentally friendly alternatives. The ban spurred innovation within the solvent industry, emphasizing the development of sustainable and less harmful substitutes. Trichloroethylene, for example, experienced a resurgence in certain applications such as metal degreasing, though it also carries its own set of environmental and health concerns.
The Legacy of 1,1,1-Trichloroetano
The story of 1,1,1-trichloroetano serves as a crucial case study in the evolution of industrial chemistry and environmental regulation. It underscores the importance of continuously evaluating the environmental and health impacts of chemicals, even those initially perceived as relatively benign. While its widespread use is a thing of the past, the lessons learned from its phase-out continue to guide the development and application of solvents and other industrial chemicals, pushing the industry towards a more sustainable and responsible future. The search for truly environmentally friendly and safe alternatives remains a priority, ensuring that future generations don't face similar challenges with the chemicals we use today.
The provided text describes 1,1,1-Trichloroethane (methyl chloroform), not 1,1,2-Trichloroethane. The FAQ below addresses 1,1,1-Trichloroethane based on the provided information. Note that I cannot create an FAQ about 1,1,2-Trichloroethane because the provided text does not contain information about it.
Frequently Asked Questions about 1,1,1-Trichloroethane
What is 1,1,1-Trichloroethane?
1,1,1-Trichloroethane, also known as methyl chloroform, is a colorless, sweet-smelling liquid chloroalkane with the chemical formula CH₃CCl₃. It was widely used as a solvent before its regulation due to its excellent solvency properties and relatively low toxicity compared to other similar compounds.
How is 1,1,1-Trichloroethane produced?
Industrial production typically involves a two-step process: vinyl chloride reacts with hydrogen chloride to form 1,1-dichloroethane, which then reacts with chlorine under UV light to yield 1,1,1-trichloroethane. A less common method involves the reaction of 1,1-dichloroethene and hydrogen chloride. Stabilizers are often added to the final product to prevent degradation and metal corrosion.
What were the common uses of 1,1,1-Trichloroethane before its regulation?
Its widespread use included cleaning metal parts and circuit boards, serving as a photoresist solvent in electronics, use as an aerosol propellant and cutting fluid additive, and in inks, paints, adhesives, and coatings. It was also used in dry cleaning leather and suede, photographic film cleaning, as a thinner in correction fluids, and even explored (but not widely adopted) as an anesthetic and veterinary anthelmintic.
Why was 1,1,1-Trichloroethane regulated?
The Montreal Protocol phased out 1,1,1-trichloroethane because of its ozone-depleting properties. Although less impactful than some other ozone depleting substances due to its relatively short atmospheric lifetime (around 5 years), it's still a potent greenhouse gas (GWP of 169 relative to CO₂).
What are the health risks associated with 1,1,1-Trichloroethane?
While relatively less toxic than some other chlorinated hydrocarbons, 1,1,1-trichloroethane is a central nervous system depressant. High concentrations can cause dizziness, confusion, unconsciousness, and even death. Prolonged skin contact can cause irritation. The International Agency for Research on Cancer classifies it as a Group 2A probable carcinogen.
What replaced 1,1,1-Trichloroethane?
The ban led to a resurgence in the use of trichloroethylene for metal degreasing, and other solvents are now used in various applications. The need for environmentally friendly and safe alternatives continues to drive research and development within the solvent industry.
Is 1,1,1-Trichloroethane still used today?
Due to its ozone-depleting properties and health concerns, the use of 1,1,1-trichloroethane is significantly restricted or banned in many parts of the world. Its presence today is likely limited to remaining stockpiles or niche applications where no suitable replacements exist.