Benzyl Alcohol CAS 100-51-6: A Deep Dive into This Versatile Compound

Understanding the properties and applications of benzyl alcohol, particularly its anhydrous form with CAS registry number 100-51-6, is crucial for those in various scientific and industrial fields. This article aims to provide a comprehensive overview of this important chemical compound.
The Chemical Structure and Properties of Benzyl Alcohol CAS 100-51-6
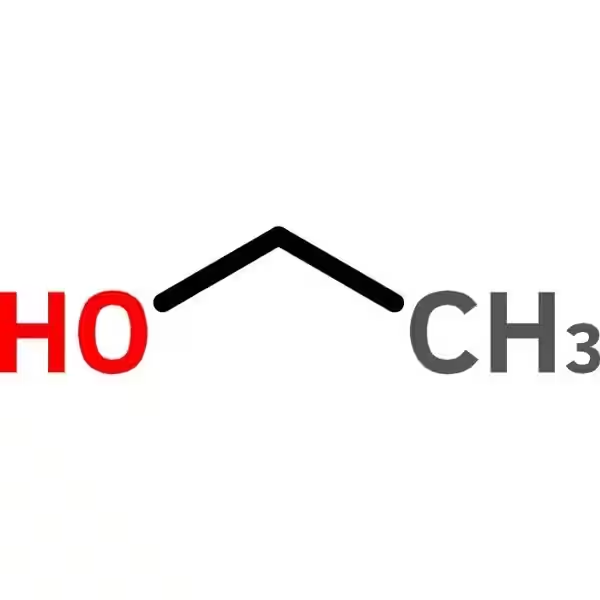
Benzyl alcohol anhydrous (99.8%, CAS 100-51-6) is a colorless liquid with a distinct aromatic odor. Its chemical formula, C₇H₈O, reflects its classification as a monoaromatic primary alcohol. This seemingly simple structure belies its remarkable versatility.
The presence of a single benzene ring attached to a hydroxyl (-OH) group is key to its unique properties. This combination creates an amphipathic molecule—meaning it possesses both hydrophobic (water-repelling) and hydrophilic (water-attracting) characteristics. This dual nature allows benzyl alcohol to dissolve a wide array of substances, making it an excellent solvent in various applications.
The classification of benzyl alcohol as a primary alcohol is equally important. The -OH group attached to a primary carbon atom grants it high reactivity, making it a valuable intermediate in a multitude of chemical syntheses. This ability to easily undergo chemical transformations contributes significantly to its use in manufacturing processes across diverse industries.
Intramolecular Hydrogen Bonding and Conformational Preference
Benzyl alcohol's ability to form an intramolecular OH…π hydrogen bond is another crucial aspect of its behavior. This internal hydrogen bonding significantly impacts its physical and chemical properties, notably influencing its conformational preference. Specifically, benzyl alcohol predominantly exists in a gauche cis conformation around the alcoholic group.
This specific arrangement influences its interactions with other molecules and its behavior within different environments. Understanding this conformational preference is vital for predicting its behavior in complex mixtures and during chemical reactions, enabling more precise control over its use in various processes.
Applications Across Diverse Industries
The versatility of benzyl alcohol (CAS 100-51-6) is reflected in its widespread use across numerous sectors. Its properties make it a valuable component in a wide range of products and processes.
Solvents and Intermediates in Chemical Synthesis
Its exceptional solvent properties make it ideal for dissolving both polar and nonpolar substances. This is highly advantageous in the cosmetic industry, where it acts as a solubilizer and carrier for other active ingredients. In the pharmaceutical industry, benzyl alcohol plays a critical role as an intermediate in the synthesis of numerous drugs and medications. Its reactivity allows it to be easily modified to create other valuable compounds, making it an indispensable component in the production of various therapeutic agents.
Flavor and Fragrance Applications
The pleasant, mild odor of benzyl alcohol contributes to its use in the flavor and fragrance industries. It's often used to enhance the scent and taste of consumer products, adding a desirable aromatic note to various formulations. This contributes to the creation of pleasant scents and tastes in a vast array of products, from perfumes and soaps to foods and beverages.
Other Uses: Preservative and Anesthetic
Beyond its solvent and intermediate roles, benzyl alcohol also finds applications as a preservative. Its bacteriostatic properties make it a valuable additive in intravenous medications and cosmetic formulations, preventing microbial growth and extending shelf life. Furthermore, it's used as a local anesthetic, often in combination with epinephrine. The antimicrobial properties contribute to its effectiveness in this role. It's also used in the treatment of head lice.
Safety and Toxicity Considerations
While generally considered safe at low concentrations, benzyl alcohol's toxicity profile must be carefully considered. Its metabolism to benzoic acid, which is subsequently excreted as hippuric acid, is a key factor in its overall safety. However, high concentrations can cause toxicity, potentially leading to respiratory issues and cardiovascular effects.
It is critically important to note that benzyl alcohol is toxic to neonates, linked to a condition known as gasping syndrome. Therefore, its use in products intended for newborns must be carefully evaluated and limited. While sensitization is rare, it can occur, primarily in individuals with stasis dermatitis. Despite its widespread use, there is currently no established evidence linking benzyl alcohol to carcinogenic or reproductive effects.
Conclusion: Benzyl Alcohol CAS 100-51-6 – A Versatile Compound with Broad Applications
In summary, benzyl alcohol (CAS 100-51-6) is a remarkable chemical compound whose versatility stems from its unique structural features. Its monoaromatic primary alcohol structure, coupled with its capacity for intramolecular hydrogen bonding and its preference for a gauche cis conformation, contributes significantly to its remarkable solubility, reactivity, and bioactivity. These properties underpin its wide-ranging applications as a solvent, synthetic intermediate, preservative, and even a local anesthetic, making it a crucial chemical in numerous industries. However, careful consideration of its toxicity profile, especially regarding neonates, is essential for safe handling and application. Understanding the properties and applications of benzyl alcohol is paramount for anyone working in related fields.
Benzyl Alcohol (CAS 100-51-6) FAQ
What is Benzyl Alcohol Anhydrous (CAS 100-51-6)?
Benzyl alcohol anhydrous (99.8% purity, CAS registry number 100-51-6) is a colorless liquid with a mild, pleasant odor. It's a monoaromatic primary alcohol, meaning its structure features a single benzene ring attached to a hydroxyl (-OH) group on a primary carbon. This unique structure is responsible for its versatile properties.
What are the key properties of Benzyl Alcohol?
Its monoaromatic primary alcohol structure gives benzyl alcohol a combination of hydrophobic (water-repelling) and hydrophilic (water-attracting) characteristics (amphipathic). This makes it an excellent solvent for a wide range of polar and nonpolar substances. The -OH group allows for various chemical transformations, making it a valuable intermediate in chemical synthesis. It also forms an intramolecular OH…π hydrogen bond, influencing its physical and chemical properties and contributing to its preference for a gauche cis conformation.
What are the industrial applications of Benzyl Alcohol?
Benzyl alcohol has diverse applications across various industries:
- Cosmetics: Used as a solvent, solubilizer, and carrier for active ingredients.
- Pharmaceuticals: Serves as an intermediate in the synthesis of numerous drugs and medications; also used as a preservative.
- Flavor and Fragrance: Contributes to desirable scents and tastes in consumer products.
- Other: Solvent in paints, inks, and coatings. Precursor to esters and ethers used in perfumes and flavorings.
How is Benzyl Alcohol produced?
Industrially, benzyl alcohol is primarily produced by hydrolyzing benzyl chloride (derived from toluene) or by hydrogenating benzaldehyde. Laboratory synthesis methods include Grignard reactions or the Cannizzaro reaction of benzaldehyde.
Is Benzyl Alcohol safe?
Benzyl alcohol is generally considered safe at low concentrations, with moderate acute toxicity. However, high concentrations can cause toxicity, including respiratory and cardiovascular effects. It's particularly toxic to neonates. While sensitization is rare, its metabolism to benzoic acid and subsequent excretion as hippuric acid is a key aspect of its handling in the body. Long-term carcinogenic or reproductive effects have not been established.
What are the key chemical reactions Benzyl Alcohol participates in?
Benzyl alcohol readily forms esters with carboxylic acids (benzyl esters are valuable protecting groups in organic synthesis). It also participates in reactions such as the Ritter reaction with acrylonitrile.
Why is the high purity (99.8%) of the anhydrous form important?
The high purity of the anhydrous form is crucial for demanding applications where even trace impurities could impact the final product quality or performance, particularly in pharmaceuticals and cosmetics.