Understanding Isobutyric Acid: From Rancid Butter to Biological Significance

Isobutyric acid, a compound with a somewhat controversial aroma, holds a surprisingly diverse range of applications and biological roles. Its presence in various natural sources and its industrial production methods make it a fascinating subject for exploration. This article delves into the multifaceted world of isobuteric acid, exploring its properties, production, uses, and biological significance.
The Chemistry of Isobutyric Acid
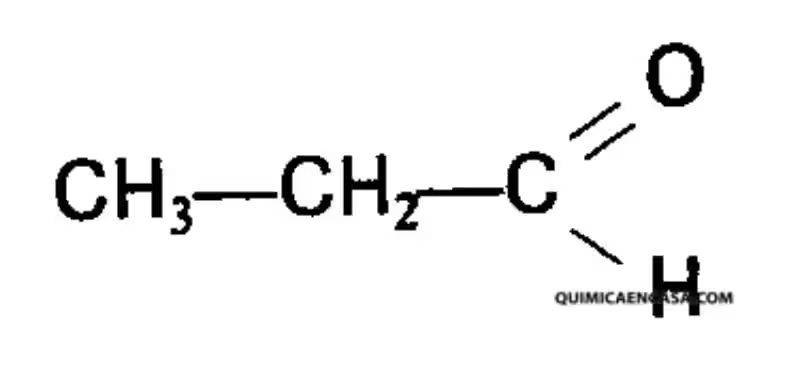
Isobutyric acid, also known as 2-methylpropanoic acid or isobutanoic acid, has the chemical formula (CH₃)₂CHCO₂H. This seemingly simple structure belies its complex behavior and diverse applications. It's a short-chain branched-chain fatty acid, a colorless liquid at room temperature.
Its distinctive feature is the presence of a methyl group (CH₃) branching off the carbon next to the carboxyl group (-COOH). This branching significantly affects its physical and chemical properties compared to its straight-chain counterpart, n-butyric acid. For example, this structural difference influences its boiling point, solubility, and reactivity. The branched structure also impacts its interaction with enzymes and receptors in biological systems.
Physical and Chemical Properties
Isobutyric acid possesses a characteristic pungent odor, frequently described as reminiscent of rancid butter, sweat, or vomit. However, it's important to note that a segment of the population is anosmic to this odor, meaning they cannot detect it. This variation in human perception highlights the subjective nature of odor descriptions. Beyond its smell, the compound is soluble in water and many organic solvents, making it versatile in both industrial and laboratory settings.
Further chemical properties include its pKa value, which indicates its acidity, and its reactivity as a typical carboxylic acid. It readily participates in reactions forming esters, amides, and other derivatives. This is crucial in understanding its various applications and roles in chemical synthesis.
Industrial Production and Synthesis of Isobutyric Acid
The industrial production of isobutyric acid predominantly relies on the oxidation of isobutyraldehyde. Isobutyraldehyde, itself a byproduct of propylene hydroformylation, undergoes a controlled oxidation process yielding the desired acid. This method is economically viable and produces large quantities of isobutyric acid.
Other synthetic routes involve the Koch reaction, a high-pressure hydrocarboxylation of propylene. This method provides an alternative pathway for the synthesis. More recently, advancements in biotechnology utilizes genetically engineered bacteria to produce isobutyric acid from readily available sugar feedstocks, providing a more sustainable method of production. This technique offers a greener alternative, reducing reliance on petrochemical-based processes.
Laboratory Synthesis Methods
Several laboratory-scale methods exist for the synthesis of isobutyric acid. These methods are often employed for research purposes or small-scale production, where industrial-scale methods are impractical. These methods include:
- Hydrolysis of isobutyronitrile: Using alkalis to break down isobutyronitrile.
- Oxidation of isobutanol: Employing potassium dichromate and sulfuric acid as oxidizing agents.
- Reaction of methacrylic acid: Utilizing sodium amalgam in the presence of proton donors.
These laboratory methods showcase the versatility of isobutyric acid synthesis, providing options depending on the availability of starting materials and desired scale.
Isobutyric Acid in Food and Biology
Isobutyric acid and its volatile esters contribute significantly to the flavor profiles of various foods. Its presence in carobs, vanilla, and Arnica dulcis root highlights its natural occurrence. Furthermore, its ethyl ester is found in croton oil. A joint FAO/WHO panel has assessed its safety as a food additive, concluding that it poses no health concerns at typical intake levels.
Biological Roles and Significance
Beyond its culinary roles, isobutyric acid holds significant biological importance. It's a component of human vaginal secretions, classified as a copulin – a short-chain fatty acid. Intriguingly, fluctuations in isobutyric acid levels during the menstrual cycle suggest a potential role as an indicator of ovulatory status, a phenomenon observed in both humans and chimpanzees. This highlights the potential of isobutyric acid as a biomarker. The metabolism of isobutyric acid in plants is also an area of ongoing research, exploring its role in plant physiology and biochemistry. This multifaceted significance underscores its importance in various biological systems.
Conclusion: The Versatility of Isobutyric Acid
Isobutyric acid, despite its pungent odor, is a versatile compound with applications ranging from industrial chemical synthesis to its role as a flavoring agent and potential biomarker. Its industrial production methods continue to evolve towards more sustainable and environmentally friendly processes, while its biological roles remain an active area of research. Understanding its properties and applications provides a valuable insight into its diverse influence in various fields.
Isobutyric Acid FAQ
What is isobutyric acid?
Isobutyric acid, also known as 2-methylpropanoic acid or isobutanoic acid, is a short-chain branched-chain fatty acid with the chemical formula (CH₃)₂CHCO₂H. It's a colorless liquid with a distinctive, unpleasant odor often described as rancid butter or sweat, although some individuals cannot detect this smell.
What is the odor of isobutyric acid like?
Many describe the odor of isobutyric acid as rancid butter or a similar unpleasant, pungent smell. However, it's important to note that a portion of the population is anosmic to it, meaning they cannot smell it.
Where is isobutyric acid found naturally?
Isobutyric acid occurs naturally in several sources, including carob beans, vanilla, Arnica dulcis root, and as an ethyl ester in croton oil. It's also a minor product of human gut microbiome activity and can be generated through the metabolism of food-derived esters.
How is isobutyric acid produced industrially?
The primary industrial method involves the oxidation of isobutyraldehyde, a byproduct of propylene hydroformylation. Other methods include the high-pressure hydrocarboxylation (Koch reaction) of propylene or using genetically modified bacteria fed with sugars.
What are some laboratory synthesis methods for isobutyric acid?
Laboratory synthesis can involve the hydrolysis of isobutyronitrile using alkalis, the oxidation of isobutanol with potassium dichromate and sulfuric acid, or the reaction of methacrylic acid with sodium amalgam in the presence of proton donors.
How does isobutyric acid react chemically?
Isobutyric acid behaves like a typical carboxylic acid, readily forming amides, esters, anhydrides, and chlorides. Its acid chloride is a common intermediate for synthesizing these derivatives. Oxidation reactions yield different products depending on the oxidizing agent; for example, chromic acid oxidation produces acetone, while alkaline potassium permanganate oxidation yields α-hydroxyisobutyric acid.
Is isobutyric acid safe for consumption?
A joint FAO/WHO panel has assessed isobutyric acid's safety as a food additive and concluded that it poses no concerns at typical intake levels. It's naturally present in many foods and contributes to their flavor profiles.
What is the biological significance of isobutyric acid?
Isobutyric acid is a component of human vaginal secretions, belonging to a group of short-chain fatty acids called copulins. Fluctuations in its levels during the menstrual cycle suggest a potential role as an indicator of ovulatory status, a phenomenon also observed in chimpanzees. Its metabolism has also been studied in plants.
What are the main uses of isobutyric acid?
Isobutyric acid and its derivatives have applications in the food industry (flavoring agent), the production of various chemicals, and as intermediates in organic synthesis. Its esters are used in fragrances and flavorings. It also has biological relevance, as noted above.
Is isobutyric acid soluble in water?
Yes, isobutyric acid is soluble in water and also in various organic solvents.