Understanding Hydroxypropyl Acrylate: A Versatile Monomer

Hydroxypropyl acrylate (HPA) is a fascinating chemical compound with a wide range of applications. Its unique chemical structure allows for diverse reactivity, making it a valuable building block in the creation of various polymers and materials. However, due to its inherent reactivity and toxicity, understanding its properties and handling precautions is crucial. This article will explore the key features of HPA, its reactivity, its applications, and the safety measures necessary for its handling.
The Chemical Structure of Hydroxypropyl Acrylate and its Significance
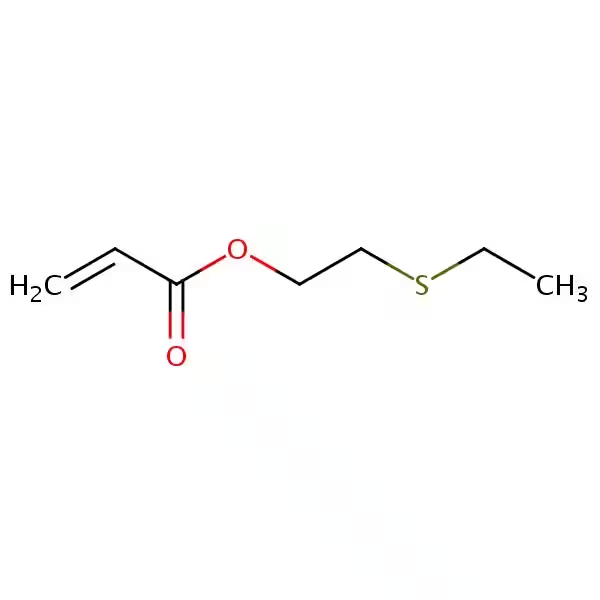
The chemical structure of hydroxypropyl acrylate is the key to understanding its versatility. It possesses two distinct functional groups: a vinyl group (characteristic of acrylates) and a hydroxyl group (-OH). This dual functionality is what sets HPA apart and enables its participation in a variety of chemical reactions.
The vinyl group allows HPA to readily undergo polymerization reactions, forming long chains of repeating HPA units. This polymerization can occur on its own (homopolymerization) or with other vinyl monomers (copolymerization). Copolymerization is particularly important, allowing for the fine-tuning of polymer properties by combining HPA with other monomers possessing desired characteristics, such as increased strength, flexibility, or water resistance.
The hydroxyl group adds another dimension to HPA's reactivity. This group can participate in reactions with a variety of other functional groups, expanding HPA's application beyond simple vinyl polymerization. For example, it can react with isocyanates to form polyurethane linkages, with anhydrides to form esters, and with epoxides through ring-opening reactions. These reactions lead to the creation of diverse materials with a broad spectrum of properties.
Applications of Hydroxypropyl Acrylate
The diverse reactivity of hydroxypropyl acrylate translates into a wide array of applications across numerous industries. The versatility of HPA allows for the development of materials with tailored properties, making it a vital component in various products.
Polyurethane Synthesis
The hydroxyl group in HPA readily reacts with di- or polyisocyanates, which is a cornerstone of polyurethane synthesis. This reaction creates urethane linkages, resulting in polyurethane-based materials. These materials are ubiquitous, finding applications in:
- Coatings: Providing durable, protective finishes on various surfaces.
- Adhesives: Creating strong bonds between different materials.
- Elastomers: Producing flexible and resilient materials.
- Foams: Forming lightweight and insulating materials.
The specific properties of the resulting polyurethane depend heavily on the choice of isocyanate and reaction conditions.
Polyester and Epoxy-Based Materials
HPA's hydroxyl group also enables its incorporation into polyester and epoxy-based materials. Reactions with anhydrides produce polyester polymers, commonly used in:
- Textiles: Confers desirable properties such as softness and durability.
- Coatings: Provides protective and aesthetic qualities.
- Plastics: Contributes to material strength and flexibility.
Similarly, reactions with epoxides via ring-opening create modified epoxy materials. By incorporating HPA, the resulting epoxy-based materials can exhibit improved flexibility or other characteristics dependent on the specifics of the reaction.
Safety Considerations and Handling of Hydroxypropyl Acrylate
Hydroxypropyl acrylate presents both health and reactivity hazards. It’s crucial to handle HPA with appropriate safety measures in place to mitigate these risks.
HPA is a hazardous substance, toxic by inhalation and skin absorption. It causes severe irritation and burns to the eyes and skin, and inhalation can lead to lung injury. Ingestion causes mouth and stomach irritation. Therefore, personal protective equipment (PPE) including gloves, eye protection, lab coat, and respiratory protection is essential when handling HPA.
Furthermore, HPA is prone to exothermic polymerization, especially under conditions of heat or contamination. This polymerization can generate significant pressure, potentially leading to container rupture. Proper storage conditions, avoiding heat sources and contaminants, are crucial. In case of spills, immediate action is required to isolate the area and absorb the spilled HPA with inert materials. Furthermore, appropriate emergency response procedures should be established and personnel trained in their implementation.
The 200 ppm of hydroquinone monomethyl ether (HQME) present in the specific formulation (CAS number 25584-83-2) acts as an inhibitor, preventing premature polymerization during storage and transport. This inhibitor is essential for maintaining the stability and usability of the HPA.
Conclusion: The Versatility and Importance of Hydroxypropyl Acrylate
Hydroxypropyl acrylate, with its unique dual functionality, stands as a versatile monomer with widespread applications. Its ability to participate in various polymerization and modification reactions allows for the creation of materials with tailored properties. Understanding the chemical structure of HPA and its reactivity is paramount to leveraging its potential effectively and safely. However, its inherent reactivity and toxicity necessitate strict adherence to safety protocols during handling, storage, and disposal to ensure worker safety and prevent incidents. The importance of HPA in various industries underscores its significance as a key building block in polymer chemistry and material science.
Hydroxypropyl Acrylate (HPA) FAQ
What is Hydroxypropyl Acrylate (HPA)?
Hydroxypropyl acrylate (HPA) is a clear, colorless liquid with the chemical formula C₆H₁₀O₃ and a molecular weight of 130. It's a mixture of isomers and contains 200 ppm of hydroquinone monomethyl ether (HQME) as a polymerization inhibitor. Its CAS number is 25584-83-2. HPA is a versatile monomer used in various industrial applications due to its unique chemical structure.
What makes HPA so versatile?
HPA possesses both a vinyl group (characteristic of acrylates) and a hydroxyl group. This dual functionality allows for a wide range of chemical reactions and polymerizations. The vinyl group enables copolymerization with other vinyl monomers, creating materials with tailored properties. The hydroxyl group allows reactions with di- or polyisocyanates, anhydrides, and epoxides, leading to polyurethane, polyester, and epoxy-based materials respectively.
What are the applications of HPA?
Because of its versatility, HPA is used as a building block in many polymers and is found in various applications across numerous industries. These include coatings, adhesives, elastomers, foams, textiles, and plastics. The specific application depends on the choice of co-monomers and the type of reaction used.
Why does HPA contain Hydroquinone Monomethyl Ether (HQME)?
The 200 ppm of HQME acts as a polymerization inhibitor. This prevents HPA from undergoing spontaneous polymerization during storage and handling, ensuring its stability and extending its shelf life. Without the inhibitor, the HPA would likely polymerize, rendering it unusable.
What are the safety concerns associated with HPA?
HPA is classified as a hazardous substance. It is toxic via inhalation and skin absorption, causing severe irritation and burns. Inhalation can lead to lung injury, and ingestion causes mouth and stomach irritation. HPA can also undergo exothermic polymerization, potentially causing container rupture. Appropriate personal protective equipment (PPE), including gloves, apron, boots, and respiratory protection, is essential when handling HPA. Spills should be handled with extreme caution, and appropriate emergency procedures should be followed.
What is the NFPA 704 rating for HPA?
The NFPA 704 diamond for HPA indicates a serious health hazard (3), moderate instability (2), and low flammability (1).
What first aid measures should be taken in case of HPA exposure?
In case of inhalation, move the affected person to fresh air. For skin or eye contact, flush thoroughly with water. For ingestion, seek medical advice immediately. In all cases, seek immediate medical attention.
What are the recommended methods for handling HPA spills?
Isolate the spill area. Eliminate all ignition sources. Absorb spills with inert materials. Prevent entry into confined spaces. Use appropriate PPE. Consult the Safety Data Sheet (SDS) for detailed spill response procedures.
What is the role of the vinyl group in HPA?
The vinyl group allows HPA to participate in copolymerization reactions with other vinyl monomers, enabling the creation of polymers with a wide range of properties.
What is the role of the hydroxyl group in HPA?
The hydroxyl group allows HPA to react with various functional groups, such as isocyanates (forming urethanes), anhydrides (forming esters), and epoxides (via ring-opening), expanding the range of possible applications and polymer types.
What happens if HPA is heated?
Heating HPA can cause exothermic polymerization, potentially leading to container rupture and the release of toxic and corrosive fumes.
Can HPA react with metals?
Contact with certain metals may generate flammable hydrogen gas.
What is the specific gravity of HPA?
The specific gravity of HPA is approximately 1.06, meaning it is denser than water.