Understanding Acetonitrilo: A Deep Dive into its Properties, Applications, and Safety

Acetonitrilo, also known as acetonitrile (CH₃CN or MeCN), is a fascinating chemical compound with a wide range of applications. It's the simplest organic nitrile, a colorless liquid with unique properties that make it indispensable in various industries and research settings. This article will explore acetonitrilo in detail, covering its production, properties, applications, and safety considerations.
The Chemistry of Acetonitrilo: Structure and Properties
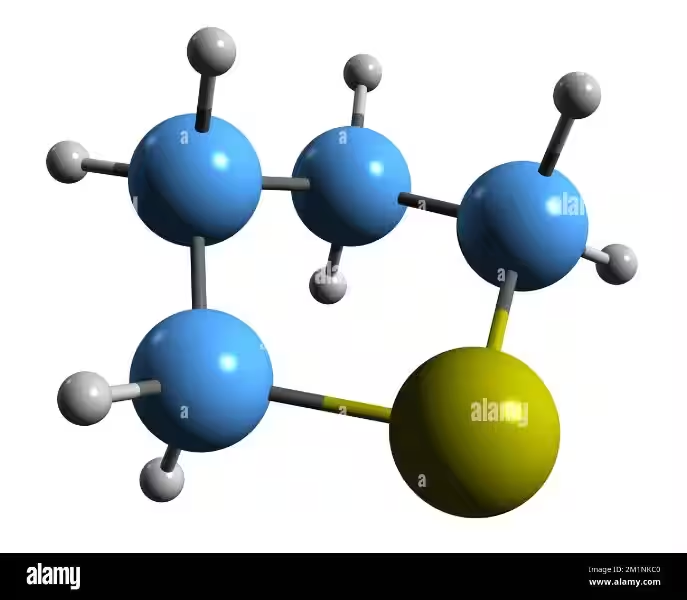
Acetonitrilo boasts a simple yet significant chemical structure. Its linear N≡C−C skeleton features a notably short carbon-nitrogen triple bond. This structure gives rise to a number of key properties:
A crucial aspect of acetonitrilo is its high dielectric constant, approximately 38.8. This property refers to a substance's ability to reduce the force between oppositely charged particles within it. This high dielectric constant makes acetonitrilo an excellent solvent for many ionic compounds.
It also possesses a significant dipole moment of 3.92 D, indicating a substantial charge separation within the molecule. This contributes to its ability to dissolve both polar and nonpolar substances, making it a versatile solvent. Furthermore, acetonitrilo exhibits UV transparency, a desirable characteristic for spectroscopic applications. Its relatively low viscosity and chemical inertness further enhance its usefulness in various processes.
Acetonitrilo's Production and Availability
The primary method for acetonitrilo production is as a byproduct of acrylonitrile manufacturing through propylene ammoxidation. This process involves the reaction of propylene, ammonia, and oxygen under specific conditions. Acetonitrile is thus largely tied to the production levels of acrylonitrile.
This dependency can lead to fluctuations in acetonitrilo availability. For example, the significant acetonitrile shortage observed between 2008 and 2009 was directly linked to decreased acrylonitrile production due to the global economic downturn, the Beijing Olympics (which impacted Chinese production), and Hurricane Ike’s damage to a US plant. This highlights the interconnectedness of global chemical markets and the contingent nature of acetonitrile supply.
The Versatile Uses of Acetonitrilo: From Industry to Lab
The unique properties of acetonitrilo translate into a wide array of applications across several sectors.
Acetonitrilo in Industrial Processes
In the petroleum industry, acetonitrilo plays a crucial role in the purification of butadiene. Its selective absorption of butadiene from hydrocarbon mixtures makes it an essential component of refinery processes. This showcases acetonitrilo's ability to separate specific components within complex mixtures.
Beyond refineries, acetonitrilo finds use in other industrial settings. Its high dielectric constant and ability to dissolve electrolytes contribute to its utility in battery technologies. The solvent's properties are ideal for improving battery performance.
Acetonitrilo in the Laboratory
The laboratory is where acetonitrilo truly shines. Its medium polarity and aprotic nature make it an excellent solvent for many chemical reactions and analytical techniques.
It's a popular choice as a mobile phase in high-performance liquid chromatography (HPLC) and liquid chromatography-mass spectrometry (LC-MS). Its low viscosity and chemical inertness minimize interference with the analytical process, leading to precise and accurate results. In addition, acetonitrilo is frequently employed in oligonucleotide synthesis, a critical process in molecular biology and biotechnology.
Acetonitrilo in Organic and Inorganic Synthesis
Acetonitrilo acts as a valuable two-carbon building block in organic synthesis. It is a precursor to various chemicals, including acetamidine hydrochloride, thiamine, and 1-naphthaleneacetic acid. These applications demonstrate acetonitrile's versatility as a reagent in the creation of more complex molecules.
In inorganic chemistry, acetonitrilo's nitrogen atom possesses a lone electron pair, enabling it to form complexes with transition metals. This property has led to its use as a ligand in various metal-catalyzed reactions. A notable example includes the synthesis of bis(acetonitrile)palladium dichloride, a useful palladium complex in many catalytic cycles.
Safety Considerations and Handling of Acetonitrilo
While widely used, acetonitrilo presents some safety concerns. Its moderate toxicity stems from its metabolism into hydrogen cyanide (HCN), a highly toxic substance. Although its metabolism is relatively slow, delayed onset of symptoms (2-12 hours) is possible.
These symptoms can range from mild (such as nausea and vomiting) to severe (including convulsions, coma, and respiratory failure). Appropriate safety measures, including adequate ventilation and personal protective equipment (PPE), are crucial when handling acetonitrilo. The use of acetonitrilo in cosmetics is banned in several regions, such as the European Economic Area, due to these toxicity concerns.
Treatment for acetonitrilo poisoning involves administering oxygen and using specific antidotes like sodium nitrite and sodium thiosulfate, following established cyanide poisoning treatment protocols. This highlights the importance of immediate medical attention in case of accidental exposure.
Conclusion: The Importance of Acetonitrilo
Acetonitrilo, despite its inherent toxicity, remains a crucial chemical compound with diverse applications across various fields. Its unique properties, including its high dielectric constant, dipole moment, and UV transparency, make it a valuable solvent in numerous industrial and laboratory settings. Understanding its properties, applications, and safety considerations is vital for anyone working with or around this versatile chemical. Proper handling and safety precautions are paramount to mitigating any potential risks associated with its use. The continuing demand for acetonitrilo reflects its indispensable role in modern chemistry and technology.
Acetonitrile (CH₃CN) Frequently Asked Questions
What is acetonitrile?
Acetonitrile (CH₃CN), also known as methyl cyanide or MeCN, is the simplest organic nitrile. It's a colorless liquid primarily produced as a byproduct of acrylonitrile manufacturing. Its chemical structure features a linear N≡C−C skeleton with a short C≡N triple bond. It possesses a high dielectric constant and dipole moment, making it a versatile solvent in various applications.
What are the key physical and chemical properties of acetonitrile?
Acetonitrile is a colorless liquid with a high dielectric constant (approximately 38.8), a significant dipole moment (3.92 D), and UV transparency. It's miscible with water and many organic solvents but immiscible with saturated hydrocarbons. Its low viscosity and chemical inertness contribute to its usefulness in many applications. It has a linear molecular structure.
What are the primary industrial uses of acetonitrile?
A major industrial use is in butadiene purification within refineries, where it selectively extracts butadiene from hydrocarbon mixtures. It also finds application in the manufacturing of pharmaceuticals and photographic films.
How is acetonitrile used in laboratories?
In laboratories, acetonitrile serves as a medium-polarity, aprotic solvent. Its properties make it suitable for high-performance liquid chromatography (HPLC), liquid chromatography-mass spectrometry (LC-MS), oligonucleotide synthesis, battery applications, and cyclic voltammetry.
What is acetonitrile’s role in organic synthesis?
In organic synthesis, acetonitrile acts as a two-carbon building block for the production of various chemicals, including acetamidine hydrochloride, thiamine, and 1-naphthaleneacetic acid. Its nitrogen atom's lone pair allows it to form complexes with transition metals. It can also form adducts with Lewis acids.
How is acetonitrile produced?
The dominant method for acetonitrile production is as a byproduct of acrylonitrile production via the ammoxidation of propylene. While other synthetic routes exist, they are not currently commercially viable.
What happened during the acetonitrile shortage of 2008-2009?
A significant acetonitrile shortage occurred between 2008 and 2009 due to reduced acrylonitrile production resulting from the global economic slowdown, the 2008 Beijing Olympics (which impacted Chinese production), and Hurricane Ike (which damaged a US plant). Since acetonitrile is a byproduct of acrylonitrile production, this decrease in acrylonitrile manufacturing directly affected acetonitrile availability.
What are the safety concerns associated with acetonitrile?
Acetonitrile exhibits moderate toxicity. Its metabolism produces hydrogen cyanide (HCN), the source of its toxic effects. While its metabolism is relatively slow, delayed onset of symptoms (2-12 hours) is possible. These symptoms can include breathing difficulties, slow pulse, nausea, vomiting, and in severe cases, convulsions, coma, and respiratory failure. Treatment involves cyanide poisoning protocols. Due to its toxicity, its use in cosmetics has been banned in some regions.
How is acetonitrile metabolized and excreted?
Acetonitrile is metabolized relatively slowly into hydrogen cyanide. Excretion occurs primarily through exhalation and urine. The relatively slow metabolism allows for detoxification to thiocyanate and excretion before significant cyanide buildup occurs.
What are the major global producers of acetonitrile?
Major global producers include companies like INEOS, DuPont, Carlo Erba Reagents, Panreac, and J.T. Baker Chemical, although the specific market share of each can fluctuate.
Is acetonitrile flammable?
Yes, acetonitrile is flammable. Appropriate safety precautions should be taken when handling and storing it.
What is the typical purity of commercially available acetonitrile?
Commercially available acetonitrile is available in various grades, with high-purity grades exceeding 99.9% purity common. The required purity depends on the intended application.
Can acetonitrile be recycled or recovered?
Yes, acetonitrile can be recycled and recovered in some industrial applications through distillation or other purification methods. However, the feasibility depends on the level of contamination and the specific process.
What is the difference between acetonitrile and other nitriles?
Acetonitrile is the simplest organic nitrile. Compared to other nitriles, its relatively slow metabolism to hydrogen cyanide contributes to its lower overall acute toxicity.
Does acetonitrile react with bases or acids?
Acetonitrile is relatively inert to many bases, but it can react with strong acids and strong bases under certain conditions. Specific reactions depend on the strength and conditions of the reaction.
What are the storage recommendations for acetonitrile?
Acetonitrile should be stored in a cool, dry, well-ventilated area, away from incompatible materials and ignition sources. Properly sealed containers should be used to prevent evaporation and contamination.
Is acetonitrile harmful to the environment?
Acetonitrile can be harmful to aquatic life. Proper disposal methods should be followed to minimize environmental impact.
What first aid measures should be taken in case of acetonitrile exposure?
In case of acetonitrile exposure, immediately remove the affected person from the source of exposure and seek medical attention. Fresh air and oxygen are crucial. Do not induce vomiting.
What are the regulatory aspects surrounding the use of acetonitrile?
Regulations concerning the use and handling of acetonitrile vary by region. Consult local and national regulations for specific guidelines. Its use in cosmetics is banned in the European Economic Area.
What are some alternative solvents to acetonitrile?
Depending on the application, alternative solvents include methanol, ethanol, tetrahydrofuran (THF), or dimethylformamide (DMF). The choice of alternative depends on the specific requirements of the application.